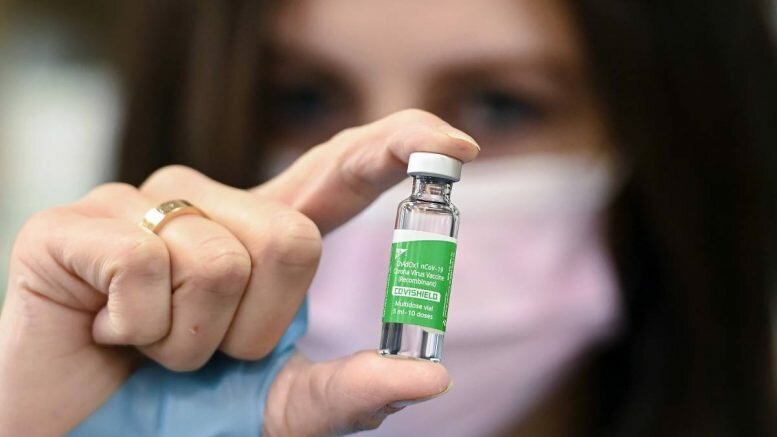
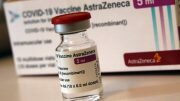
Vaccine manufacturer AstraZeneca writes on Saturday night that analyses of their coronavirus vaccine have not shown signs of an increased risk of blood clots or a reduction in platelets.
The Saturday statement from press manager Christina Malmberg Hëgerstrand in AstraZeneca noted that the company has performed an analysis of safety data after more than 17 million vaccine doses have been administered.
These have not shown signs of an increased risk of blood clots in the legs or lungs or a reduction in the number of platelets in the blood.
The statement from AstraZeneca comes after the National Institute of Public Health (FHI) and the Norwegian Medicines Agency convened an emergency press conference on Saturday in the wake of three health workers being admitted to Rikshospitalet with a blood clot after being vaccinated with a vaccine from AstraZeneca.
“In fact, the reported numbers of these types of events for AstraZeneca’s COVID-19 vaccine are not greater than the number that would have occurred naturally in the unvaccinated population,” Hëgerstrand wrote in the statement.
The company has started a review of all safety data.
It also pointed out that the European Medicines Agency (EMA) has also asked other manufacturers of the COVID-19 vaccine to assess the occurrence of reduction in platelets.
Source: © NTB Scanpix / #Norway Today / #NorwayTodayNews
Do you have a news tip for Norway Today? We want to hear it. Get in touch at [email protected]



Be the first to comment on "AstraZeneca rejects claims that its vaccine increases the risk of blood clots"